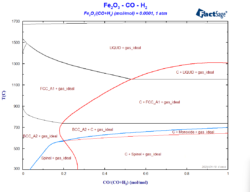
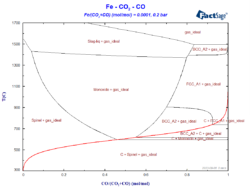
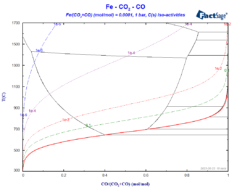
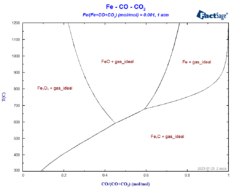
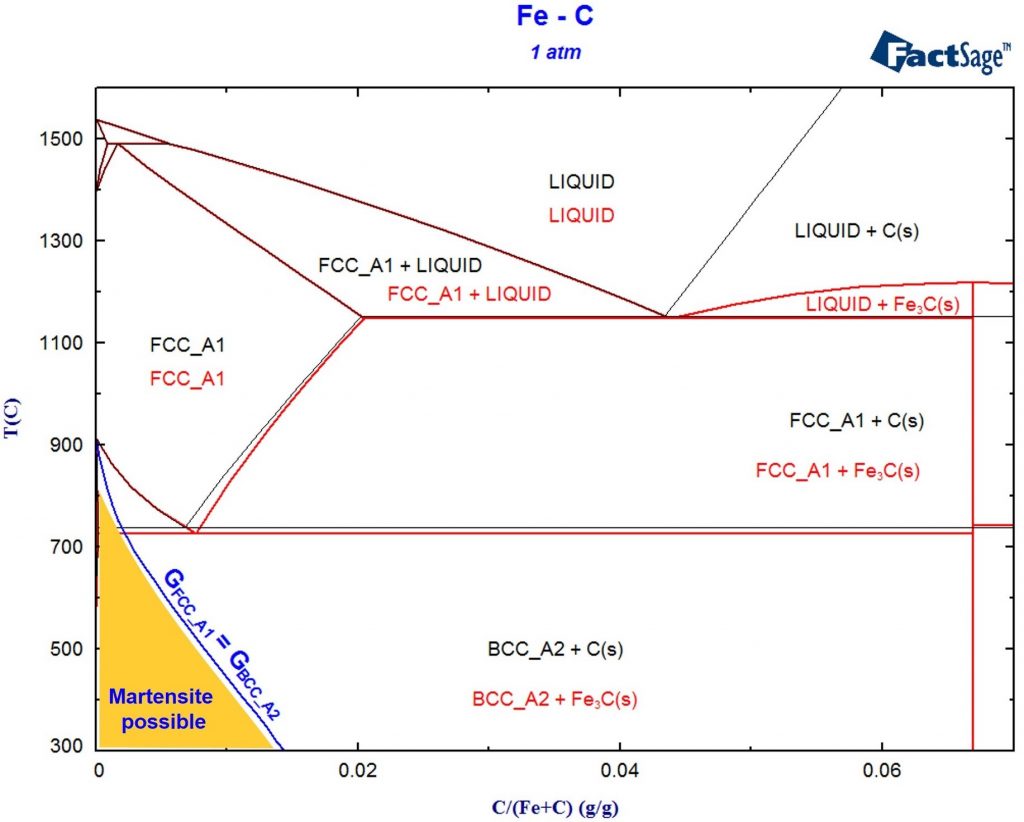
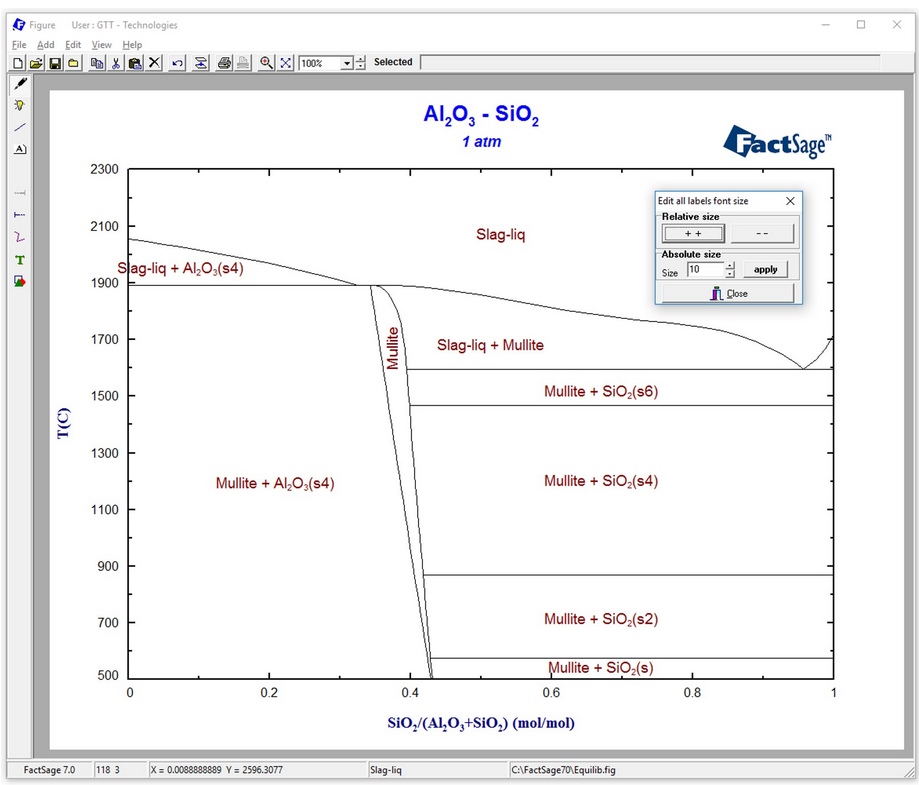
Constructing a Blast Furnace Diagram with FactSage – Part IV
In order to reduce the CO2 emissions caused by the iron & steel industry, there have been initiatives in the last decades to substitute reductants based on carbon by the use of H2 as a reducing agent. These initiatives go from the direct injection of H2 at the tuyeres of the blast furnace [1, 2] […]
Constructing a Blast Furnace Diagram with FactSage – Part IV Read More »